Detection of alkaline phosphatase activity and inhibition with fluorescent hydroxyapatite nanoparticles
Abstract
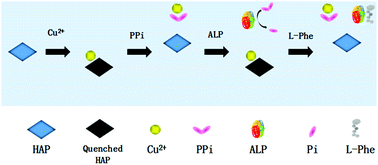
Alkaline phosphatase (ALP) is a membrane-bound glycoprotein widely distributed in humans. It can catalyze the hydrolysis of phosphate monoesters under alkaline conditions. Since the variation of ALP activity is associated with various diseases, it is crucial to develop sensitive and accurate methods for monitoring ALP activity. We report here a fluorescence assay for testing ALP activity and inhibition based on fluorescent hydroxyapatite nanoparticles (HAP-NPs). The fluorescence intensity of the HAP-NPs can be initially quenched by Cu(II) and then recovered by inorganic pyrophosphate (PPi), a substrate of ALP. So ALP can regulate the HAP-NP fluorescence intensity by hydrolyzing PPi into phosphate ions (Pi). According to the test results, ALP can be quantitatively detected in the range from 1 U L−1 to 625 U L−1. When applied for real sample analysis, the level of ALP activity in human serum samples detected by our assay was in good agreement with the clinical test results. In addition, the assay was utilized for screening ALP inhibitors. These data indicate that the assay has good potential for clinical and pharmaceutical applications.


 Please wait while we load your content...
Please wait while we load your content...