A superior dye adsorbent towards the hydrogen evolution reaction combining active sites and phase-engineering of (1T/2H) MoS2/α-MoO3 hybrid heterostructured nanoflowers†
Abstract
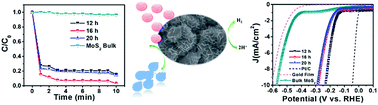
Here, we demonstrate the successful synthesis of (1T/2H) MoS2/α-MoO3 heterostructured nanoflowers at a low temperature of 200 °C by a one-step hydrothermal method. By tuning the reaction time under the influence of thiourea and hydrazine hydrate, we established a complete phase-engineered MoS2 with 1T and 2H phases on the surface of α-MoO3. Active sites associated with the phase-engineered (1T/2H) MoS2/α-MoO3 hybrid nanoflowers enable them to exhibit dual roles as a superior dye adsorbent and an electrocatalyst towards the hydrogen evolution reaction. The 2H-rich (1T/2H) MoS2/α-MoO3 hybrid heterostructured nanoflowers prepared at 16 h achieved a high surface area of 37.97 m2 g−1, and 97% of the RhB dye with an initial concentration of 47.9 mg L−1 was removed within 10 min through the adsorption process, which is the highest known removal efficiency reported in the literature. As a hydrogen evolution reaction (HER) electrocatalyst in acidic solution, the 1T-rich (1T/2H) MoS2/α-MoO3 hybrid heterostructured nanoflowers prepared at 12 h exhibited a highly efficient catalytic activity by achieving a low overpotential of 232 mV at a current density of 10 mA cm−2, which is comparable to those of previously reported HER catalysts based on MoS2. Moreover, this sample reached a low Tafel slope of 81 mV dec−1 and was stable when operated for more than 1000 cycles.


 Please wait while we load your content...
Please wait while we load your content...