Mutually beneficial Co3O4@MoS2 heterostructures as a highly efficient bifunctional catalyst for electrochemical overall water splitting†
Abstract
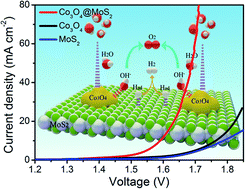
Designing low-cost and highly efficient bifunctional electrocatalysts for compatible integration with the hydrogen evolution reaction (HER) and the oxygen evolution reaction (OER) for overall water splitting is critical but challenging. Herein, mutually beneficial Co3O4@MoS2 heterostructures were adopted to efficiently balance both HER and OER performance by improving the sluggish kinetics. These heterostructures synergistically favoured the reduction of the energy barrier of the initial water dissociation step and optimization of the subsequent H adsorption/desorption for MoS2 in alkaline HER. Moreover, the adsorption of oxygen intermediates was enhanced for Co3O4 in the OER process. As a result, the Co3O4@MoS2 heterostructures showed excellent overall water splitting performance with a low overpotential and Tafel slope.


 Please wait while we load your content...
Please wait while we load your content...