Facile and template-free fabrication of mesoporous 3D nanosphere-like MnxCo3−xO4 as highly effective catalysts for low temperature SCR of NOx with NH3†
Abstract
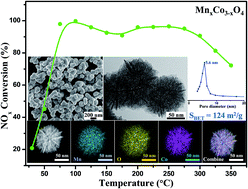
Mesoporous 3D MnxCo3−xO4 nanospheres with an open structure were fabricated through a template-free and facile approach. The MnxCo3−xO4 achieved a specific surface area as high as 143.7 m2 g−1. The process was systematically studied via TEM, HRTEM, EDS, ICP, XRD, TG, pH and FTIR. A two-step formation mechanism, namely the formation of CoOOH and MnO2 by a redox reaction between KMnO4 and Co(NO3)2·6H2O, followed by a calcination process in air, was proposed. The synthesis conditions including the reaction duration, reaction temperature, reactant concentration, feeding ratio, calcination temperature and the types of cobalt precursor used were also carefully investigated. This indicates that the approach is universal, green, facile, controllable and effective. The MnxCo3−xO4 catalysts exhibited outstanding performance in the application of low-temperature selective catalytic reduction of NOx with NH3. A wide working temperature window of 75 to 325 °C (NOx conversion above 80%) was achieved. The good performance benefited from the high BET surface area, a strong adsorption capacity, abundant acid sites, robust redox properties, abundant oxygen vacancies and metal–metal interactions in MnxCo3−xO4.


 Please wait while we load your content...
Please wait while we load your content...