Tuning the emission of a water-soluble 3-hydroxyflavone derivative by host–guest complexation†
Abstract
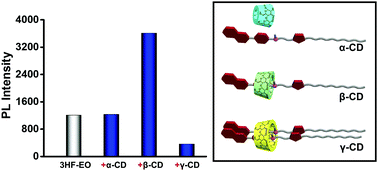
3-Hydroxyflavone derivatives have great potential as fluorescent probes for bio-labeling in aqueous medium. They were extensively studied in various organic solvents for the “excited state intramolecular proton transfer” process, but seldom addressed in aqueous solution due to the poor water solubility. Herein, an amphiphilic molecule bearing 3-hydroxyflavone and oligo(ethylene oxide) (denoted as 3HF-EO) was designed and synthesized. Different from the fluorescence in organic solvents, 3HF-EO in aqueous solution showed a remarkable single fluorescence emission, which is ascribed to the fluorescence of its anionic species. We found that the fluorescence intensity could be efficiently tuned via host–guest complexation. α-CD has little effect on the emission, while β-CD and γ-CD lead to enhanced and reduced emissions of 3HF-EO, respectively. The 1H NMR and 2D NOESY NMR spectra indicate that α-CD barely had any interaction with 3HF-EO, while β-CD and γ-CD formed complexes with one and two 3HF-EO molecules, respectively. These results provide a sound explanation for the modulated fluorescence intensity.


 Please wait while we load your content...
Please wait while we load your content...