Stereoselective cobalt-catalyzed halofluoroalkylation of alkynes†
Abstract
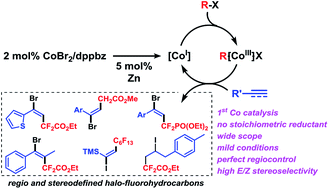
Stereoselective additions of highly functionalized reagents to available unsaturated hydrocarbons are an attractive synthetic tool due to their high atom economy, modularity, and rapid generation of complexity. We report efficient cobalt-catalyzed (E)-halofluoroalkylations of alkynes/alkenes that enable the construction of densely functionalized, stereodefined fluorinated hydrocarbons. The mild conditions (2 mol% cat., 20 °C, acetone/water, 3 h) tolerate various functional groups, i.e. halides, alcohols, aldehydes, nitriles, esters, and heteroarenes. This reaction is the first example of a highly stereoselective cobalt-catalyzed halo-fluoroalkylation. Unlike related cobalt-catalyzed reductive couplings and Heck-type reactions, it operates via a radical chain mechanism involving terminal halogen atom transfer which obviates the need for a stoichiometric sacrificial reductant.


 Please wait while we load your content...
Please wait while we load your content...