Role of enhanced solubility in esterification of 2,5-furandicarboxylic acid with ethylene glycol at reduced temperatures: energy efficient synthesis of poly(ethylene 2,5-furandicarboxylate)†
Abstract
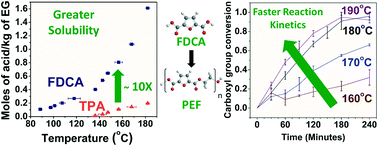
Poly(ethylene 2,5-furandicarboxylate) (PEF) has garnered considerable industrial and academic interest as a renewable alternative to traditional polyesters due to its superior barrier and thermal properties. While efforts for the industrial scale production of PEF from 2,5-furandicarboxylic acid (FDCA) and ethylene glycol (EG) are underway, most of the published literature on PEF follows the conventional terephthalic acid (TPA) based polyester synthesis protocol. In this study, we reveal for the first time that the solubility of FDCA is an order of magnitude higher in EG compared to that in TPA at the process temperatures. The enhanced solubility of FDCA in EG results in improved esterification kinetics especially at temperatures from 180–210 °C to yield complete end group conversion. We further demonstrate that it is advantageous to perform the direct esterification step of PEF synthesis in a lower temperature range than that in previous reports reducing the potential side reactions in addition to cost and energy savings at the industrial scale. This work can provide new insights into the sustainable synthesis of FDCA-based polyesters for bio-based packaging.


 Please wait while we load your content...
Please wait while we load your content...