Direct synthesis of acrylate monomers in heterogeneous continuous flow processes†
Abstract
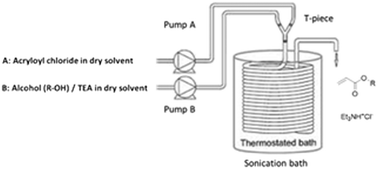
An efficient, safe and convenient laboratory scale continuous flow process for synthesis of (meth)acrylate monomers is described. (Meth)acryloyl chloride is reacted with n-octanol and other alcohols in the presence of triethyl amine in a tubular reactor, resulting in excellent conversions of alcohols to their corresponding esters within 0.5 to 5 minutes of reaction times. The formation of an unwanted side-compound, 3-chloropropionyl ester, is significantly minimized to less than 1% and the formed slurry can be processed without difficulty using an ultrasonication-assisted flow strategy. The synthesis is carried out over a period of 4 h without clogging or pressure build-up, with an operation throughput of 78.6 g h−1 of n-octyl acrylate monomer on laboratory scale, with 95% isolated yield. This procedure provides an efficient alternative synthetic route of functional (meth)acrylate esters, minimizes the side product and facilitates the handling of the formed slurry in the process. Synthesis of various (meth)acrylates is demonstrated, including biobased terpenyl acrylates. This development will play an important role for an on-demand production of (meth)acrylate monomers in fast, reliable and energy-efficient process.


 Please wait while we load your content...
Please wait while we load your content...