Polymer semiconductors incorporating head-to-head linked 4-alkoxy-5-(3-alkylthiophen-2-yl)thiazole†
Abstract
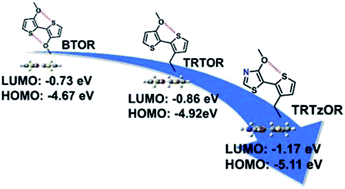
Head-to-head linked bithiophenes with planar backbones hold distinctive advantages for constructing organic semiconductors, such as good solubilizing capability, enabling narrow bandgap, and effective tuning of frontier molecular orbital (FMO) levels using minimal thiophene numbers. In order to realize planar backbone, alkoxy chains are typically installed on thiophene head positions, owing to the small van der Waals radius of oxygen atom and accompanying noncovalent S⋯O interaction. However, the strong electron donating alkoxy chains on the electron-rich thiophenes lead to elevated FMO levels, which are detrimental to material stability and device performance. Thus, a new design approach is needed to counterbalance the strong electron donating property of alkoxy chains to bring down the FMOs. In this study, we designed and synthesized a new head-to-head linked building block, 4-alkoxy-5-(3-alkylthiophen-2-yl)thiazole (TRTzOR), using an electron-deficient thiazole to replace the electron-rich thiophene. Compared to previously reported 3-alkoxy-3′-alkyl-2,2′-bithiophene (TRTOR), TRTzOR is a weaker electron donor, which considerably lowers FMOs and maintains planar backbone through the noncovalent S⋯O interaction. The new TRTzOR was copolymerized with benzothiadiazoles with distinct F numbers to yield a series of polymer semiconductors. Compared to TRTOR-based analogous polymers, these TRTzOR-based polymers have broader absorption up to 950 nm with lower-lying FMOs by 0.2–0.3 eV, and blending these polymers with PC71BM leads to polymer solar cells (PSCs) with improved open-circuit voltage (Voc) by ca. 0.1 V and a much smaller energy loss (Eloss) as low as 0.59 eV. These results demonstrate that thiazole substitution is an effective approach to tune FMO levels for realizing higher Vocs in PSCs and the small Eloss renders TRTzOR a promising building block for developing high-performance organic semiconductors.


 Please wait while we load your content...
Please wait while we load your content...