Studies on structure-function relationships of acetolactate decarboxylase from Enterobacter cloacae
Abstract
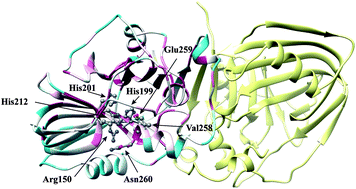
Acetoin is an important bio-based platform chemical with wide applications. Among all bacterial strains, Enterobacter cloacae is a well-known acetoin producer via α-acetolactate decarboxylase (ALDC), which converts α-acetolactate into acetoin and is identified as the key enzyme in the biosynthetic pathway of acetoin. In this work, the enzyme properties of Enterobacter cloacae ALDC (E.c.-ALDC) were characterized, revealing a Km value of 12.19 mM and a kcat value of 0.96 s−1. Meanwhile, the optimum pH of E.c.-ALDC was 6.5, and the activity of E.c.-ALDC was activated by Mn2+, Ba2+, Mg2+, Zn2+ and Ca2+, while Cu2+ and Fe2+ significantly inhibited ALDC activity. More importantly, we solved and reported the first crystal structure of E.c.-ALDC at 2.4 Å resolution. The active centre consists of a zinc ion coordinated by highly conserved histidines (199, 201 and 212) and glutamates (70 and 259). However, the conserved Arg150 in E.c.-ALDC orients away from the zinc ion in the active centre of the molecule, losing contact with the zinc ion. Molecular docking of the two enantiomers of α-acetolactate, (R)-acetolactate and (S)-acetolactate allows us to further investigate the interaction networks of E.c.-ALDC with the unique conformation of Arg150. In the models, no direct contacts are observed between Arg150 and the substrates, which is unlikely to maintain the stabilization function of Arg150 in the catalytic reaction. The structure of E.c.-ALDC provides valuable information about its function, allowing a deeper understanding of the catalytic mechanism of ALDCs.


 Please wait while we load your content...
Please wait while we load your content...