Synthesis of multifunctional CuFe2O4–reduced graphene oxide nanocomposite: an efficient magnetically separable catalyst as well as high performance supercapacitor and first-principles calculations of its electronic structures†
Abstract
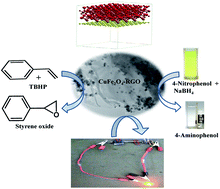
Here, we report an ‘in situ’ co-precipitation reduction based synthetic methodology to prepare CuFe2O4 nanoparticle–reduced graphene oxide (CuFe2O4–RGO) nanocomposites. First principles calculations based on Density Functional Theory (DFT) were performed to obtain the electronic structures and properties of CuFe2O4, graphene and CuFe2O4–graphene composites, and to understand the interfacial interaction between CuFe2O4 and graphene in the composite. The synergistic effect, which resulted from the combination of the unique properties of RGO and CuFe2O4 nanoparticles, was exploited to design a magnetically separable catalyst and high performance supercapacitor. It has been demonstrated that the incorporation of RGO in the composite enhanced its catalytic properties as well as supercapacitance performance compared with pure CuFe2O4. The nanocomposite with 96 wt% CuFe2O4 and 4 wt% RGO (96CuFe2O4–4RGO) exhibited high catalytic efficiency towards (i) reduction of 4-nitrophenol to 4-aminophenol, and (ii) epoxidation of styrene to styrene oxide. For both of these reactions, the catalytic efficiency of 96CuFe2O4–4RGO was significantly higher than that of pure CuFe2O4. The easy magnetic separation of 96CuFe2O4–4RGO from the reaction mixture and good reusability of the recovered catalyst also showed here. 96CuFe2O4–4RGO also demonstrated better supercapacitance performance than pure CuFe2O4. 96CuFe2O4–4RGO showed specific capacitance of 797 F g−1 at a current density of 2 A g−1, along with ∼92% retention for up to 2000 cycles. To the best of our knowledge, this is the first investigation on the catalytic properties of CuFe2O4–RGO towards the reduction of 4-nitrophenol and the epoxidation reaction, and DFT calculations on the CuFe2O4–graphene composite have been reported.


 Please wait while we load your content...
Please wait while we load your content...