Environment-friendly magnetic Fe–Ce–W catalyst for the selective catalytic reduction of NOx with NH3: influence of citric acid content on its activity-structure relationship†
Abstract
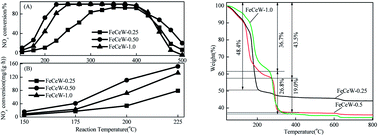
The influence of the citric acid content on the structural and redox properties of a magnetic iron–cerium–tungsten mixed oxide catalyst prepared through a microwave-assisted citric acid sol–gel method is investigated via TG–DTG–DSC, XRD, N2 adsorption–desorption, XPS, H2-TPR and NH3-TPD. Additionally, the NH3-SCR activity of the magnetic FeCeW-m (m = 0.25, 0.5 and 1.0) catalysts are also studied. The results indicate that an increase in citric acid content strengthens the sol–gel reaction between citric acid and metal ions and promotes the formation of the γ-Fe2O3 crystallite not α-Fe2O3. Meanwhile, it decreases the BET surface area and pore volume of the catalyst. Furthermore, the surface concentration of iron species on the catalyst is enhanced when the molar ratio of citric acid/(Fe + Ce + W) increases from 0.25 to 1.0, but its surface absorbed oxygen and total oxygen concentration decrease. The magnetic FeCeW-0.5 catalyst shows the best reducibility at temperatures below 790 °C. The increase in the citric acid content inhibits the formation of acid sites in the catalyst, thus the magnetic FeCeW-0.25 catalyst possesses the most Lewis acid sites and Brønsted acid sites among the catalysts. The enhancement in citric acid content is beneficial to improve the SCR reaction rates normalized by the surface area of the catalyst. This catalyst exhibits high anti-SO2 and H2O poisoning, and the molar ratio of citric acid/(Fe + Ce + W) affects the adsorption of NOx species on its surface.


 Please wait while we load your content...
Please wait while we load your content...