Towards selective electrochemical conversion of glycerol to 1,3-propanediol†
Abstract
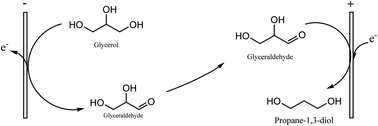
1,3-propanediol (1,3-PD) is a bulk chemical with myriad applications in polymers, lubricants, cosmetics, foods industries and in the synthesis of heterocyclic compounds. Current commercial production of 1,3-PD involves a thermocatalytic process using acrolein (DuPont) and ethylene oxide (Shell) as starting feedstock. These feedstocks are petroleum-based and there are many efforts at using glycerol as low cost biomass-derived feedstock for 1,3-PD production. A number of catalyst designs and bacterial & fungal strains are being explored for respective catalytic and fermentation routes to glycerol-to-1,3-PD. However, the electrochemical method received little attention for the purpose. In this work, in order to explore the possibility of using partly refined glycerol byproduct of biodiesel production as feedstock, we investigated conversion and 1,3-PD selectivity of glycerol electrolysis in chloride media. We demonstrated selective glycerol-to-1,3-PD conversion using Pt or RuO2-based dsa as anode and Zn or Pb as cathode in NaCl and KCl at pH 1. This electrochemical glycerol-to-1,3-PD conversion is not only green, it is a potential process network loop between biodiesel production and chlor-alkali industry.


 Please wait while we load your content...
Please wait while we load your content...