Investigation of ultra-low friction on steel surfaces with diketone lubricants
Abstract
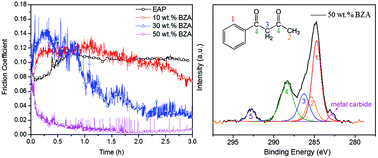
In this study, the lubricating properties of ethylacetophenone (EAP) solutions with different benzoylacetone (BZA) concentrations on steel surfaces were investigated. The results indicate that with an increase in the BZA concentration in the solution, the friction coefficient decreases, and an ultra-low friction coefficient (μ ≈ 0.005–0.008) is obtained for the solution with 50 wt% BZA concentration. This demonstrates that both the applied normal load and the sliding velocity have significant influence on the realization of ultra-low friction for the 50 wt% BZA solution. Furthermore, the chemical states of the friction surfaces and the components of the 50 wt% BZA solution were detected via X-ray photoelectron spectroscopy (XPS) and infrared spectroscopy (IR), respectively. The analyses reveal that a tribochemical reaction occurs between the BZA molecules and the rubbing surfaces, and the chelate not only can disperse in the solution, but can also form chemical adsorption layers on the rubbing surfaces. In addition, the mechanism of ultra-low friction has been discussed based on the results of these analyses. Both the influence of the hydrodynamic effect and the existence of chemically absorbed films on the steel surfaces lead to a reduction in the friction coefficient. This study reveals that diketone is a promising lubricant for ultralow friction and has great potential in industrial applications.


 Please wait while we load your content...
Please wait while we load your content...