Double B←N bridged bipyridine-containing polymer acceptors with enhanced electron mobility for all-polymer solar cells†
Abstract
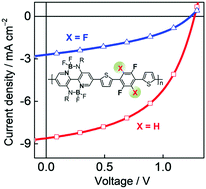
With the aim of developing polymer electron acceptors with high electron mobilities (μe) for all-polymer solar cells (all-PSCs), we synthesized two novel polymer acceptors (P-BNBP-2f and P-BNBP-4f) containing alternate double B←N bridged bipyridine (BNBP) and 2,2′-(2,5-difluoro-1,4-phenylene)dithiophene/2,2′-(perfluoro-1,4-phenylene)dithiophene with extended conjugated structures. In comparison to P-BNBP-4f, P-BNBP-2f exhibits a small π–π stacking distance of 3.60 Å, leading to a sufficient electron mobility of 5.40 × 10−4 cm2 V−1 s−1 (measured by the space-charge-limited current method). This μe value is among the highest values of the conventional polymer acceptors, and is close to the hole mobilities (μh) of high-efficiency polymer donors. Moreover, P-BNBP-2f possesses high-lying LUMO/HOMO energy levels of −3.42/−5.81 eV, which match well with that of the reported excellent polymer donors of 2D-conjugated bithienyl-benzodithiophene-alt-fluorobenzotriazole (J61) and thienyl-substituted BDT with alkoxycarbonyl-substituted thieno[3,4-b]thiophene (PBDTTT-E-T). In addition, the blend films based on P-BNBP-2f exhibit intermixed morphologies, which are beneficial for efficient excition dissociation. As a result, all-PSCs with P-BNBP-2f as an electron acceptor work very well and exhibit a power conversion efficiency of 5.46%. These results demonstrate that high electron mobility of a polymer electron acceptor is very important to produce efficient all-PSCs.

 Please wait while we load your content...
Please wait while we load your content...