Ultralong needle-like N-doped Co(OH)F on carbon fiber paper with abundant oxygen vacancies as an efficient oxygen evolution reaction catalyst†
Abstract
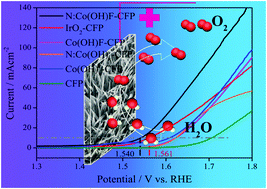
Although significant efforts have been devoted to designing efficient electrocatalysts for the oxygen evolution reaction, further progress is still needed regarding modulating the microstructures of electrocatalysts to further improve catalytic activity. Herein, utilizing ionic compound NaF and heteroatom-N-rich urea as morphology directing reagents, ultralong needle-like N-doped Co(OH)F supported on carbon fiber paper has been synthesized through a one-step hydrothermal method. Specifically, the hydrothermal kettle provides a stable environment for the formation of ultra-long needle-like structures. Meanwhile, with the addition of halogen anion F− and heteroatom N, a wealth of oxygen vacancies are obtained, and the OER activity is enhanced. The as-prepared N-doped Co(OH)F-CFP (N:Co(OH)F-CFP) has excellent hydrophilicity, which facilitates its contact with the electrolyte and then accelerates the passing of electrons back and forth between the surface of the material and the electrolyte. Under alkaline conditions, N:Co(OH)F-CFP's OER activity is superior to that of IrO2. Its potential can reach 1.540 V (vs. RHE) at 10 mA cm−2 with a Tafel slope of 69.75 mV dec−1 in 1 M KOH solution.


 Please wait while we load your content...
Please wait while we load your content...