Aqueous cationic homo- and co-polymerizations of β-myrcene and styrene: a green route toward terpene-based rubbery polymers†
Abstract
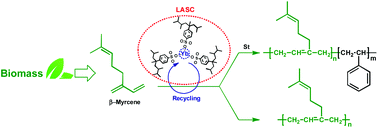
Aqueous cationic polymerization of β-myrcene, a naturally occurring monomer, was performed for the first time using a water-dispersible Lewis acid surfactant combined catalyst (LASC) prepared from ytterbium chloride and a branched sodium dodecylbenzenesulfonate surfactant. At 40 °C, poly(β-myrcene)s of high molecular weight (Mn up to 150 kg mol−1) and low glass transition temperature (Tg < −63 °C) were obtained. They possess predominantly 1,4-units (∼43% cis and ∼50% trans) and few 3,4-units (∼7%) together with both regular head-to-tail (62–72%) and inverse (5–10% head-to-head and 23–28% tail-to-tail) sequences. The LASC showed also high efficiency in catalyzing the emulsion cationic copolymerization of β-myrcene and styrene, affording the synthesis of random copolymers with high molecular weights (Mn from 60 to 120 kg mol−1) and single glass transition temperatures (Tg's from −43 to 15 °C). Finally, a simple procedure was developed to recover the catalyst from the reaction mixture, which showed high activity in the β-myrcene emulsion cationic polymerization even after five successive cycles of extraction.


 Please wait while we load your content...
Please wait while we load your content...