Scandium-catalyzed copolymerization of myrcene with ethylene and propylene: convenient syntheses of versatile functionalized polyolefins†
Abstract
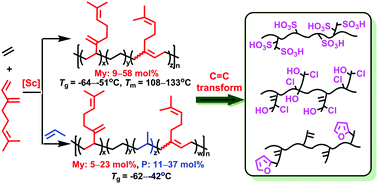
The copolymerization of myrcene with ethylene and propylene using half-sandwich scandium complexes bearing various auxiliary ligands has been examined. In the copolymerization of myrcene with ethylene, the THF-free aminobenzyl scandium complex bearing a relatively small cyclopentadienyl ligand (C5H5)Sc(CH2C6H4NMe2-o)2 (1) afforded diblock myrcene–ethylene copolymers with a high myrcene content and predominant cis-1,4-structure, whereas the analogous scandium complex with a larger cyclopentadienyl ligand (C5Me4SiMe3)Sc(CH2C6H4NMe2-o)2 (2) afforded multiblock myrcene–ethylene copolymers with different myrcene contents and a predominant 3,4-structure. In contrast, the THF-containing trimethylsilylmethyl scandium complex (C5Me4SiMe3)Sc(CH2SiMe3)2(THF) (3) afforded alternating myrcene–ethylene copolymers on using a sufficient amount of myrcene. More remarkably, the terpolymerization of myrcene with ethylene and propylene has also been realized using a scandium complex 3, yielding a new family of myrcene-based ethylene–propylene–diene rubbers (EPDM) with versatile C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds in the main and side chains. By efficiently transforming the C
C double bonds in the main and side chains. By efficiently transforming the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C double bonds of the resulting myrcene-based EPDM into other polar groups, versatile functionalized polyolefins such as thioacetic acid, sulfoacid, and epoxidized, hydroxylated–chlorinated and furan functionalized polyolefins were obtained.
C double bonds of the resulting myrcene-based EPDM into other polar groups, versatile functionalized polyolefins such as thioacetic acid, sulfoacid, and epoxidized, hydroxylated–chlorinated and furan functionalized polyolefins were obtained.


 Please wait while we load your content...
Please wait while we load your content...