Modulated photochemical reactivities of O-acetylated (3′,5′-dimethoxyphenyl)heteroaryl acyloin derivatives under direct irradiation and photo-induced electron transfer conditions†
Abstract
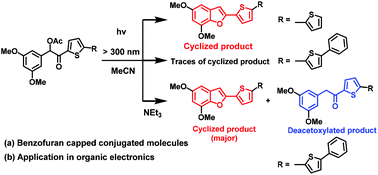
3′,5′-Dimethoxybenzoin esters are important photoremovable protecting groups which form 2-phenylbenzofuran derivatives upon photo-release. We utilized a similar concept to test a photochemical method of installing a benzofuran moiety to the conjugated backbone by subjecting O-acetylated (3′,5′-dimethylphenyl)heteroaryl acyloin derivatives through direct photo irradiation and a photo-induced electron transfer reaction. These photochemical methods were explored for a variety of heteroaromatic substrates appended on the ketone part of the O-acetylated cross-acyloin derivatives. The furan, thiophene and bithiophene derivatives led to the expected cyclized (benzofuran capped) products but the derivatives with extended conjugation decomposed under direct irradiation. However, under irradiation in the presence of an electron donor such as triethylamine, the extended acyloin derivatives afforded both cyclized and deacetoxylated products. The semiconducting nature of the extended cyclized products was also explored and tested for solution-processed organic field effect transistors, providing a maximum hole mobility of 1.3 × 10−6 cm2 V−1 s−1.


 Please wait while we load your content...
Please wait while we load your content...