Chemoselective synthesis of isolated and fused fluorenones and their photophysical and antiviral properties†
Abstract
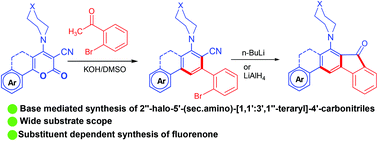
Highly functionalized fluorenones were synthesized by an intramolecular cyclization of 2′′-halo-[1,1′:3′,1′′-terphenyl]-4′-carbonitriles in the presence of n-butyllithium or lithium aluminium hydride. The precursor was synthesized by ring transformation of 2-oxo-6-aryl/heteroaryl-4-(sec.amino)-2H-pyran-3-carbonitriles or 2-oxobenzo[h]chromenes with o-bromo/chloro/fluoro-acetophenone under basic conditions in moderate yield. We performed the control experiment to understand the proposed mechanism and found that the presence of a secondary amine in the starting material directs the reactivity. The photophysical properties of 3-methoxy-7-(piperidin-1-yl)-5H-indeno[2,1-b]phenanthren-8(6H)-one was explored and solvent dependent emission was observed. These compounds were also tested against HIV-1 and low to moderate activity was observed.
- This article is part of the themed collection: Synthetic methodology in OBC


 Please wait while we load your content...
Please wait while we load your content...