Investigations into the biosynthesis of the antifungal strobilurins†
Abstract
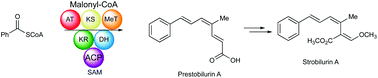
The strobilurins are important antifungal metabolites isolated from a number of basidiomycetes and have been valuable leads for the development of commercially important fungicides. Isotopic labelling studies with early and advanced intermediates confirm for the first time that they are produced via a linear tetraketide, primed with the rare benzoate starter unit, itself derived from phenylalanine via cinnamate. Isolation of a novel biphenyl metabolite, pseudostrobilurin B, provides evidence for the involvement of an epoxide in the key rearrangement to form the β-methoxyacrylate moiety essential for biological activity. Formation of two bolineol related metabolites, strobilurins Y and Z, also probably involves epoxide intermediates. Time course studies indicate a likely biosynthetic pathway from strobilurin A, with the simplest non-subsubstituted benzoate ring, to strobilurin G with a complex dioxepin terpenoid-derived substituent. Precursor-directed biosynthetic studies allow production of a number of novel ring-halogenated analogues as well as a new pyridyl strobilurin. These studies also provide evidence for a non-linear biosynthetic relationship between strobilurin A and strobilurin B.
- This article is part of the themed collection: Chemical Biology in OBC


 Please wait while we load your content...
Please wait while we load your content...