Single-ligand exchange on an Au–Cu bimetal nanocluster and mechanism†
Abstract
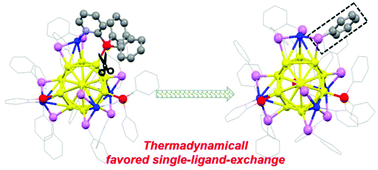
An Au–Cu bimetallic nanocluster co-capped by selenolate and phosphine is obtained and its X-ray structure shows an icosahedral Au13 kernel surrounded by three CuSe2PPh2Py motifs and one CuSe3 motif, formulated as [Au13Cu4(PPh2Py)3(SePh)9]. Interestingly, a single-ligand exchange process is observed in the growth reaction, in which an [Au13Cu4(PPh2Py)4(SePh)8]+ intermediate is first formed, but a prolonged reaction leads to one PPh2Py ligand being selectively replaced by a PhSe-ligand. DFT simulations reveal that both steric hindrance and bond dissociation energy have great effects on the single-ligand exchange reaction as well as the thermodynamics, which help to understand the mechanism of the ligand exchange. Temperature-dependent UV-vis absorption and photoluminescence (PL) properties of the Au–Cu nanocluster imply that the optical properties are mainly contributed by the metal core. Femtosecond time-resolved pump–probe analysis maps out further details of the PL process.


 Please wait while we load your content...
Please wait while we load your content...