Natural products from thioester reductase containing biosynthetic pathways
Abstract
Covering: up to 2018
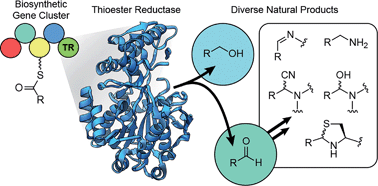
Thioester reductase domains catalyze two- and four-electron reductions to release natural products following assembly on nonribosomal peptide synthetases, polyketide synthases, and their hybrid biosynthetic complexes. This reductive off-loading of a natural product yields an aldehyde or alcohol, can initiate the formation of a macrocyclic imine, and contributes to important intermediates in a variety of biosyntheses, including those for polyketide alkaloids and pyrrolobenzodiazepines. Compounds that arise from reductase-terminated biosynthetic gene clusters are often reactive and exhibit biological activity. Biomedically important examples include the cancer therapeutic Yondelis (ecteinascidin 743), peptide aldehydes that inspired the first therapeutic proteasome inhibitor bortezomib, and numerous synthetic derivatives and antibody drug conjugates of the pyrrolobenzodiazepines. Recent advances in microbial genomics, metabolomics, bioinformatics, and reactivity-based labeling have facilitated the detection of these compounds for targeted isolation. Herein, we summarize known natural products arising from this important category, highlighting their occurrence in Nature, biosyntheses, biological activities, and the technologies used for their detection and identification. Additionally, we review publicly available genomic data to highlight the remaining potential for novel reductively tailored compounds and drug leads from microorganisms. This thorough retrospective highlights various molecular families with especially privileged bioactivity while illuminating challenges and prospects toward accelerating the discovery of new, high value natural products.


 Please wait while we load your content...
Please wait while we load your content...