Synthesis, structures, electrochemical and quantum chemical investigations of Ni(ii) and Cu(ii) complexes with a tetradentate Schiff base derived from 1-(2-thienyl)-1,3-butanedione†‡
Abstract
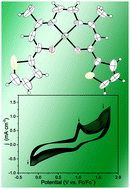
Double condensation of the newly prepared 1-(2-thienyl)-1,3-butanedione with ethylenediamine led to the formation of a novel symmetrical Schiff base proligand bearing two potentially electropolymerizable 2-thienyl groups. Both organic species were obtained in 55 and 80% yields, respectively. They exist in their respective keto–enol and enaminone tautomeric forms that were computed to be more stable by 8.6 and 30.3 kcal mol−1 than their β-diketone and keto-imine isomers. The corresponding Ni(II) and Cu(II) complexes featuring a N2O2-tetradentate Schiff base ligand were readily synthesized upon reaction of the diprotic Schiff base precursor with the appropriate hydrated metal acetates, and isolated as neutral, air and thermally stable solids in excellent yields (84 and 90%). The four compounds were characterized using various analytical and spectroscopic methods, and by an X-ray diffraction study for the two coordination complexes. Both Ni(II) and Cu(II) metal ions are four-coordinated and adopt a perfect square planar environment (τ4 values of 0.036 and 0.025) with two nitrogen and two oxygen atoms as donors. Both complexes were analyzed by cyclic voltammetry experiments showing a decrease of the current response per cycle, indicating the formation of oligomeric units. This was verified using their doping/undoping responses. The optimized geometries of the four compounds as well as the electronic structures of the two Ni(II) and Cu(II) complexes and their respective cations were analysed through DFT calculations, allowing the provision of a consistent view of their structure and properties. TDDFT calculations were used to interpret the major features of the UV-vis spectra.


 Please wait while we load your content...
Please wait while we load your content...