Supported ZnBr2 and carbon nitride bifunctional complex catalysts for the efficient cycloaddition of CO2 with diglycidyl ethers†
Abstract
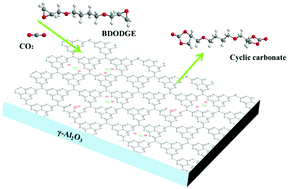
The cycloaddition reaction of carbon dioxide (CO2) and bisepoxides is an interesting route for CO2 utilization and green non-isocyanate polyurethane (NIPU) synthesis. In this work, a series of γ-Al2O3 supported ZnBr2 and carbon nitride complex catalysts (Zn–CN/γ-Al2O3) were prepared by a simple thermal polymerization method and applied to the cycloaddition reaction. The results showed that 0.05Zn–CN/γ-Al2O3 exhibited high catalytic activity, over which 91.4% 1,4-butanediol diglycidyl ether (BDODGE) conversion and 98.9% cyclic carbonate selectivity were acquired under solvent and co-catalyst free conditions. The relationship between the structural properties and catalytic performance of Zn–CN/γ-Al2O3 catalysts was studied in detail. It is found that the high surface area and the synergistic effect of Lewis acid–base duality are key factors for the high activity of catalysts. Introducing ZnBr2 could provide acid sites and induce structure change of carbon nitride, which could determine the basic intensity of catalysts. Moreover, based on the elaborate discussions, a plausible catalytic reaction mechanism was proposed.


 Please wait while we load your content...
Please wait while we load your content...