Unravelling the role of polyols with increasing carbon chain length and OH groups on the phase transition behavior of PNIPAM
Abstract
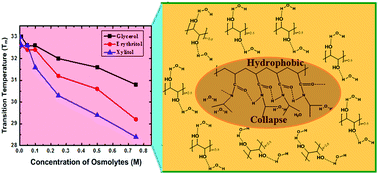
In advanced applications of pharmaceutical, agricultural and biomedical research, thermoresponsive polymers (TRPs) are potential candidates which show conformational transitions at given temperatures. The conformational transitions in TRPs depend on their intermolecular interactions with co-solvent and intramolecular interaction changes among their own segments. The transition behavior of TRPs can be altered in the presence of biocompatable osmolytes which are found to be present in all living organisms. Here, in this work, we attempted to elucidate the effect of polyols with varying C-chain length and OH groups, namely glycerol, erythritol and xylitol, on the coil-to-globule transition of poly(N-isopropylacrylamide) (PNIPAM). The results indicate that a decrease in lower critical solution temperature (LCST) of PNIPAM is observed in the presence of these additives as compared to the typical LCST of PNIPAM. The observed results may be due to the polarization ability of these osmolytes towards water molecules associated with the PNIPAM surface and so lead to hydrophobic association or agglomeration of PNIPAM that leads to solution turbidity above the specific phase transition temperature. These osmolytes promote the formation of collapsed PNIPAM at temperatures below that of the LCST of PNIPAM in a concentration dependent manner. For the current study, we employed various biophysical techniques such as UV-visible, fluorescence and Fourier-transform infrared (FTIR) spectroscopic studies along with dynamic light scattering (DLS). This work may also open new alternatives for the synthesis of PNIPAM based devices and drug delivery applications.


 Please wait while we load your content...
Please wait while we load your content...