The dual-luminescence mechanism of the ESIPT chemosensor tetrasubstituted imidazole core compound: a TDDFT study†
Abstract
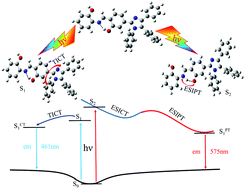
The dual-luminescence mechanism of the tetrasubstituted imidazole core (TIC) compound was theoretically explored by considering the excited-state intramolecular proton transfer (ESIPT) process in the present study. Density functional theory (DFT) and time-dependent density functional theory (TDDFT) methods have been employed to gain the geometrical conformations, electronic transitions, frontier molecular orbitals (FMOs) and various spectra in different electronic states. An intramolecular hydrogen bond O–H⋯N was observed for the TIC in the S0 state. Upon electronic excitation, the TIC molecule is predominantly excited to the S1 state as well as to an isosbestic point in the S2 state. There is a twisted intramolecular charge transfer (TICT) during the S0 → S1 (π–π*) transition and an intramolecular charge transfer (ICT) during the S0 → S2 (π–π*) transition. The intramolecular hydrogen bond becomes stronger in the S1 state than in the S0 state. This leads to a strong fluorescence emission at 461 nm. However, the calculated potential curves showed that the intramolecular hydrogen bond O–H⋯N transforms into O⋯H–N in the S2 state after the ESIPT reaction. A keto form of TIC is generated which gives rise to the other fluorescence emission at 575 nm through an ultrafast internal conversion (IC) process. This study aims to provide theoretical insights into the development of efficient chemosensors which are switched on through the ESIPT process.


 Please wait while we load your content...
Please wait while we load your content...