Reactivity of a new aryl cycloplatinated(ii) complex containing rollover 2,2′-bipyridine N-oxide toward a series of diphosphine ligands†
Abstract
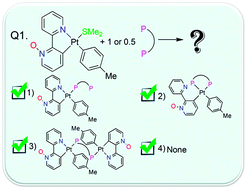
Reaction of the electron-rich complex cis-[Pt(p-Me-C6H4)2(SMe2)2] with 2,2′-bipyridine N-oxide, O-bpy occurred by rollover cyclometalation to afford complex [Pt(O-bpy)(p-Me-C6H4)(SMe2)], 1. The obtained complex was characterized using NMR spectroscopy and its solid state structure was determined by the single crystal X-ray diffraction method. The reaction of 1 with seven diphosphine ligands, 1,1-bis(diphenylphosphino)methane (dppm), 1,2-bis(diphenylphosphino)ethane (dppe), 1,3-bis(diphenylphosphino)propane (dppp), 1,4-bis(diphenylphosphino)butane (dppb), N,N-bis(diphenylphosphino)amine (dppa), 1,2-bis(diphenylphosphino)benzene (dppbz) and 1,1′-bis(diphenylphosphino)ferrocene (dppf), in different molar ratios (1 : 1 or 1 : 0.5; 1 : diphosphines) was studied. In accordance with the reaction conditions, the analogous mononuclear or binuclear diphosphine cycloplatinated complexes were yielded. The diphosphines behave as a monodentate (dppm and dppa), a bridging (dppm, dppa, dppe, dppp, dppb and dppf) or a chelated (dppe, dppp and dppbz) ligand. These behaviors depend on the bite angle of the diphosphine ligands, and the flexibility or rigidity of the alkyl and aromatic backbone between the two phosphine groups. All diphosphine platinum complexes were characterized using NMR spectroscopy and the crystal structures of some complexes were solved by X-ray diffraction.


 Please wait while we load your content...
Please wait while we load your content...