Synthesis, structures and antimicrobial activity of copper derivatives of N-substituted imidazolidine-2-thiones: unusual bio-activity against Staphylococcus epidermidis and Enterococcus faecalis†
Abstract
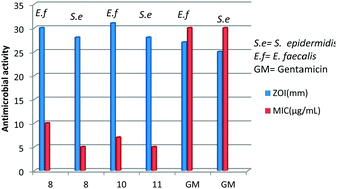
The main objective of this study was to explore the antimicrobial activity of several copper(I) complexes with N,S-donor thio-ligands against Gram positive bacteria, namely Staphylococcus aureus (MTCC 740), Staphylococcus epidermidis (MTCC 435), and Enterococcus faecalis (MTCC 439), Gram negative bacteria, Shigella flexneri (MTCC 1457) and Escherichia coli (MTCC 119), and yeast, Candida tropicalis (MTCC 230). New copper complexes were prepared from copper(I) halides and imidazolidine-2-thiones (L-NR, R = Et; Prn; Bun; Ph) with triphenylphosphine as a co-ligand. The complexes are mononuclear, [CuX(L-NR)(PPh3)2] (X, R: Cl, Prn, 1; Br, Prn, 2; Cl, Bun, 3; I, Bun, 4; I, Ph, 6) and [CuBr(L-NPh)2(PPh3)] 5, and halogen-bridged dinuclear, [Cu2(μ-X)2(L-NR)2(PPh3)2] (Br, Et, 7; Cl, Prn, 8; Br, Prn, 9; I, Prn, 10; Br, Ph, 11; I, Ph, 12). These complexes were characterized using analytical data, IR, 1H/13C-NMR spectroscopy, single crystal X-ray crystallography, electron spray ionization mass spectrometry (ESI-MS) and techniques for antimicrobial study (agar diffusion, agar dilution, MTT assay). All of the complexes are found to be bactericidal against Staphylococcus aureus. The most significant outcome of this investigation is that several complexes have shown significant activity against Staphylococcus epidermidis and Enterococcus faecalis, which is higher than that of the standard drug Gentamicin. Finally, these complexes were nearly inactive against Shigella flexneri, Escherichia coli and yeast Candida tropicalis.


 Please wait while we load your content...
Please wait while we load your content...