New insight into the effects of N^N ligand isomerization and methyl modification on the phosphorescence properties of Cu(i) complexes with (1-(2-pyridyl)pyrazole/imidazole) ligands†
Abstract
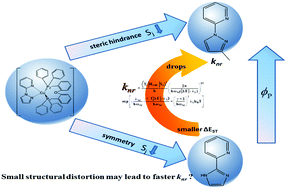
The influence of ligand isomerization and methylation modification on the phosphorescence properties of metal complexes has been attracting great interest owing to the significant improvement of phosphorescence efficiency. In recent years, experimental results have shown that the substitution of a single methyl group in the N^N ligand of a Cu(I) complex can enhance the quantum yield. To further clarify the influence of methyl substituents, we investigated a series of Cu(I) complexes with (1-(2-pyridyl) pyrazole/imidazole and bis[2-(diphenylphosphino)phenyl]ether mixed ligands)[Cu(N^N)(P^P)] based on density functional theory (DFT) and time-dependent DFT (TD-DFT) methods. The results indicate that the additional methyl could increase the ratio of the d orbital in the frontier molecular orbitals of [Cu(N^N)(P^P)], thereby enlarging the 3MLCT (%); substitution of the parazole with an imidazole group could reduce the ratio of 3MLCT involved in emission transition. Furthermore, vibration analysis reveals that a methyl group substitution in the ortho-position of N2 increases the steric hindrance, thereby suppressing the vibration of benzene and the non-radiative dissipation caused by methyl. Meanwhile, this substitution enhances the energy difference between the T1 state and the S0 state (ΔE) and reduces the non-radiative decay rate (knr). Our studies provide the intrinsic reason for the high PLQY of [Cu(N^N)(P^P)] complexes with ortho-methyl modification, including the reason for the low PLQY of the isomer.


 Please wait while we load your content...
Please wait while we load your content...