Improvement of photocatalytic activity of surfactant modified In2O3 towards environmental remediation†
Abstract
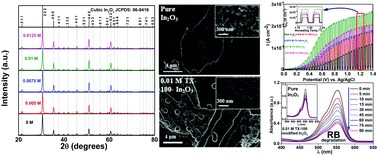
The present paper describes photocatalytically active In2O3 thin films developed using a direct drop-cast method on F-doped tin oxide (FTO) coated glass substrates using 10 mM In(NO3)3 dissolved in ethylene glycol containing 0–0.0125 M Triton-X 100 (TX-100) surfactant and annealed in air at 600 °C to obtain the desired metal oxide. The absorption spectrum measured the direct band gap of the surfactant modified In2O3 as 3.48 eV along with an indirect gap of 2.69 eV indicating near visible absorptivity of the materials, which was established through linear sweep voltammetry under periodic UV-Vis and visible irradiation for oxidation of water and a sacrificial reagent, SO32−. The electrochemical impedance spectroscopic (Mott–Schottky) analysis confirms n-type semiconductivity for these materials. Addition of an optimized level of 0.01 M TX-100 surfactant to the precursor solution improves the photoelectrochemical performance of the film to 2.3 fold. The electrochemical action spectra indicate a maximum value of the incident photon to current conversion efficiency (IPCE) for the 0.01 M surfactant modified In2O3 of 28% and the corresponding absorbed photon to current conversion efficiency (APCE) of 40%. Addition of surfactant to the In3+ precursor solution results in uniformly distributed particle growth on the surface with better crystallinity.


 Please wait while we load your content...
Please wait while we load your content...