Three in situ-synthesized novel inorganic–organic hybrid materials based on metal (M = Bi, Pb) iodide and organoamine using one-pot reactions: structures, band gaps and optoelectronic properties†
Abstract
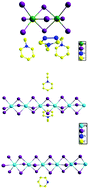
Based on metal iodide, three novel inorganic–organic hybrid materials formulated as (N-ethyl-pyridine)2(N-ethyl-1,3,4-triazole)(Bi2I9) (1), [(N-ethyl-pyridine)(PbI3)]n (2) and [(Hpyridine)(PbI3)]n (3) were in situ – synthesized solvothermally and they have been characterized by single-crystal X-ray diffraction. In compound 1, the in situ-formed (N-ethyl-pyridine)+ and (N-ethyl-1,3,4-triazole)+ cations are linked with a zero-dimensional (0D) (Bi2I9)3− cluster via strong H bonds to form a three-dimensional (3D) supramolecular architecture. While the in situ-formed (N-ethyl-pyridine)+ cations in compound 2 and (Hpyridine)+ cations in compound 3 are linked with infinite one-dimensional (1D) Pb-(μ2-I)3-Pb-(μ2-I)3-Pb chains via strong H bonds. Density functional theory (DFT) calculations demonstrate that the three compounds have different band structures. And among them, compound 1 possesses the narrowest band gap and it produces the largest photocurrent density under visible light illumination (350 nm < λ < 650 nm). Moreover, compounds 1 and 2 possess good water-proof properties in aqueous solution, which is probably due to hydrophobic aromatic cations such as (N-ethyl-pyridine)+ and (N-ethyl-1,3,4-triazole)+ in their structures.


 Please wait while we load your content...
Please wait while we load your content...