Green synthesis of high-performance LiFePO4 nanocrystals in pure water†
Abstract
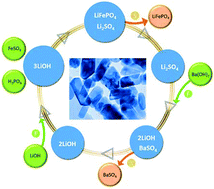
Hydrothermal synthesis of high-performance LiFePO4 nanocrystals in pure water represents a green and sustainable approach to fabricate model cathode materials for lithium ion batteries. However, fundamental and technological hurdles in terms of yield and cost remain to be overcome, though strenuous efforts have been devoted to this challenging approach. Herein, we report a microwave-assisted hydrothermal strategy that enables scalable green synthesis of high-performance LiFePO4 nanocrystals using inexpensive chemical reagents, namely, lithium hydroxide, ferrous sulfate and phosphoric acid in pure water without involving any organic solvents or surfactants. Three critical issues regarding the hurdles are simultaneously addressed. First, the dream of recycling the lithium source, the most valued reagent, in the hydrothermal synthesis comes true, which substantially reduces synthesis cost; second, to the best of our knowledge, a record high volume specific yield of more than 1.3 mol L−1 is achieved. Third, the as-synthesized phase-pure LiFePO4 nanocrystals exhibit excellent electrochemical properties including high capacity and long cyclability.


 Please wait while we load your content...
Please wait while we load your content...