Electrochemical oxidative radical oxysulfuration of styrene derivatives with thiols and nucleophilic oxygen sources†
Abstract
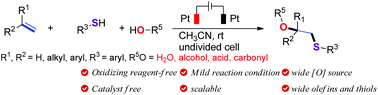
Oxydifunctionalization of olefins represents a powerful tool and yet poses a challenging task. Previous methods have usually required a stoichiometric amount of a strong oxidant and an expensive transition-metal catalyst. This work describes the first example of the electrochemical oxysulfuration reaction of olefins with thiols and nucleophilic oxygen sources. This electrochemical difunctionalization reaction is conducted under catalyst- and oxidant-free conditions, and shows good substrate generality, affording thio-substituted alcohols, ethers and γ-lactones in good chemical yields and with excellent regioselectivities. This work represents a new and green strategy for the difunctionalization of olefins, and also provides a complementary and highly valuable prospect for current methodologies for the synthesis of thio-substituted compounds.
- This article is part of the themed collection: 2018 Green Chemistry Hot Articles


 Please wait while we load your content...
Please wait while we load your content...