Compositional modification of pyrogenic products using CaCO3 and CO2 from the thermolysis of polyvinyl chloride (PVC)†
Abstract
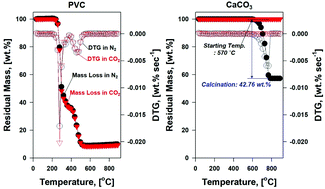
In this study, we explored the mechanistic features of CO2 in the thermolysis of PVC to modify the pyrogenic products. The mechanistic roles of CO2 in the thermolysis of PVC were varied with the pyrolytic temperature. For example, CO2 participated in unknown reactions to form CO at temperatures higher than 600 °C, where CO2 played a key role as an oxygen donor (new finding). This new thermal behavior induced by CO2 could be a new way to shift carbon from pyrolytic oil to syngas (i.e., H2 and CO) without using catalysts. This identified reaction that formed CO was noticeably enhanced in the presence of CaCO3 by up to 12 times, which was proportional to the loading of CaCO3. The stepwise thermal degradation mechanisms for PVC (i.e., dehydrogenation followed by dechlorination) provided favorable conditions for forming benzene derivatives and PAHs in pyrolytic oil. In the temperature range from 480 to 600 °C, CO2 played a key role in restricting the formation of benzene derivatives, which subsequently lowered the formation of PAHs by blocking the pathway for the gas-phase addition reactions. At the current stage of this study, the operational parameters such as the exact amount of CO2, space velocity for CO2, and other parameters for the thermolysis of PVC were fully optimized. However, all the experimental findings marginally described the possible utilization of CO2 for modifying pyrogenic products, which can be applied in various fields such as air pollutant controls (APCs) and energy applications. As such, further studies need to be conducted in the near future.


 Please wait while we load your content...
Please wait while we load your content...