Abstract
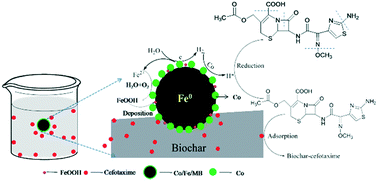
Ecological risks caused by residual antibiotics have increasingly attracted great attention. In this study, a highly efficient novel adsorbent based on Co/Fe bimetallic nanoparticles loaded with alkali modified biochar (Co/Fe/MB) was prepared to remove cefotaxime from an aqueous solution. The prepared composites were characterized with X-ray photoelectron spectroscopy (XPS), X-ray diffraction (XRD) and scanning electron microscopy (SEM). The effects of Co loading, adsorbent dosage, initial solution pH and anions (Cl−, HCO3− and EDTA) on the removal of cefotaxime were investigated. Due to the synergetic reaction of the adsorption and reduction mechanism, Co/Fe/MB revealed the highest cefotaxime removal efficiency (99.23%) in 100 min at the condition of pH = 5, T = 298 K, Co/Fe/MB dosage = 0.8 g L−1, Co loading = 0.4 wt%, cefotaxime = 20 mg L−1. The removal efficiency was further enhanced in the presence of EDTA and Cl− because of the destruction of a passivation layer on iron surface. Additionally, the degradation products and possible degradation pathway of cefotaxime by Co/Fe/MB were analyzed by high performance liquid chromatography-electrospray ionization mass spectrometry (HPLC-ESI-MS). The spectra revealed that the opening and cleavage of the β-lactam ring was the first step for cefotaxime degradation. The Co/Fe/MB composite was found to be an efficient material to remove cefotaxime from the solution.


 Please wait while we load your content...
Please wait while we load your content...