Engineering oxygen-containing and amino groups into two-dimensional atomically-thin porous polymeric carbon nitrogen for enhanced photocatalytic hydrogen production†
Abstract
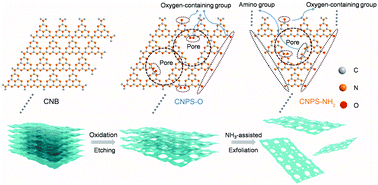
Polymeric carbon nitride (PCN) is a promising earth-abundant photocatalyst for solar energy conversion. However, the photocatalytic activities of PCN-based materials remain moderate because of their poor dispersion in water and their fast electron–hole recombination. Here, a facile two-step continuous thermal treatment strategy is presented to endow the bulk PCN nanosheets with an atomically-thin structure, strong hydrophilicity and Lewis basicity to dramatically enhance the photocatalytic hydrogen (H2) generation performance. The formation of the oxygen-containing and amino groups in the atomically-thin PCN sheets improves the charge separation and provides rich active sites for the surface reaction. Such synergistic effects lead to a superior visible-light-driven photocatalytic activity and its H2 evolution rate (1233.5 μmol h−1 g−1) is more than 11 times higher than the bulk PCN using Ni as a cocatalyst. Additionally, the H2 evolution rate can reach 20948.6 μmol h−1 g−1 using Pt as a cocatalyst under AM1.5G solar irradiation.


 Please wait while we load your content...
Please wait while we load your content...