A simple methodology for constructing ferromagnetically coupled Cr(iii) compounds†
Abstract
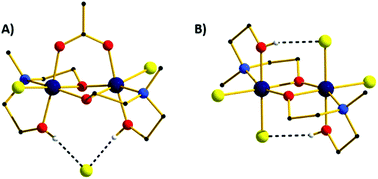
A large family of chromium(III) dimers has been synthesised and magneto-structurally characterised using a combination of carboxylate and diethanolamine type ligands. The compounds have the general formula [Cr2(R1-deaH)2(O2CR2)Cl2]Cl where R1 = Me and R2 = H (1), Me (2), CMe3 (3), Ph (4), 3,5-(Cl)2Ph (5), (Me)5Ph (6), R1 = Et and R2 = H (7), Ph (8). The compound [Cr2(Me-deaH)2Cl4] (9) was synthesised in order to study the effect of removing/adding the carboxylate bridge on the observed magnetic behaviour. Direct current (DC) magnetic susceptibility measurements showed ferromagnetic (FM) exchange interactions between the Cr(III) centres in the carboxylate bridged family with coupling constants in the range +0.37 < J < +8.02 cm−1. Removal of the carboxylate to produce the dialkoxide-bridged compound 9 resulted in antiferromagnetic (AFM) exchange between the Cr(III) ions. DFT calculations reveal the ferromagnetic exchange is the result of an orbital counter-complementarity effect occuring upon introduction of the bridging carboxylate.


 Please wait while we load your content...
Please wait while we load your content...