Superior solar-to-hydrogen energy conversion efficiency by visible light-driven hydrogen production via highly reduced Ti2+/Ti3+ states in a blue titanium dioxide photocatalyst†
Abstract
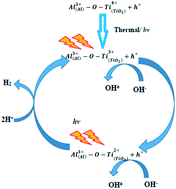
Photocatalytic water splitting is one of the most important renewable paths and a reliable hydrogen production system. In most successful molecular and supramolecular biomimetic hydrogen production methods, a photosensitizer and a catalyst were constructed where the photoexcited electron in the photosensitizer is transferred either inter- or intramolecularly to the catalytic centre. Similar to supramolecular complexes in a photocatalytic hydrogen production scheme, here we develop a redox system that contains Ti3+/Ti2+ reduced states in TiO2 which act as both visible light harvesting components and the catalytic sites for the catalytic hydrogen production with visible-near infrared photons. The Ti3+/Ti2+ states in TiO2 produce hydrogen from pure water with a solar-to-hydrogen energy conversion efficiency of 0.89% and a quantum yield of 43% at 655 nm. The mechanism of hydrogen production by the Ti3+/Ti2+ reduced states in TiO2 involves the initial generation of highly air stable and highly reduced Ti3+ and Ti2+ states in TiO2 by the formation of an AlOOH layer surrounding the anatase and rutile particles. Once Ti3+ and Ti2+ states are generated, these states are continuously self-generated via absorption of visible-near infrared radiation where hydrogen is produced by the transfer of electrons from Ti3+/Ti2+ to H+.


 Please wait while we load your content...
Please wait while we load your content...