Effect of Brønsted acid sites on the direct conversion of methane into methanol over copper-exchanged mordenite†
Abstract
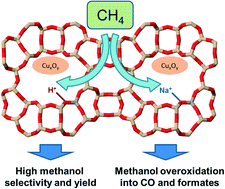
The direct conversion of methane into methanol was studied in a stepwise process over copper-exchanged mordenite. The materials were characterized by X-ray diffraction, nitrogen adsorption–desorption, FTIR of adsorbed NO and X-ray absorption spectroscopy. The methanol productivity and selectivity correlated with the amount of Brønsted acid sites in the zeolite. The in situ FTIR spectroscopy revealed that the presence of Brønsted acid sites increases the stability of formed methanol, hence avoiding its overoxidation into carbon oxides. The best performing material in terms of methanol yield was copper-exchanged mordenite with the highest amount of the Brønsted acid sites showing a methanol productivity of 118 μmol g−1 equivalent to 0.18 mol(MeOH)/mol(Cu) with 95% selectivity towards methanol.


 Please wait while we load your content...
Please wait while we load your content...