Heterogeneously catalyzed selective hydrogenation of amides to alcohols and amines†
Abstract
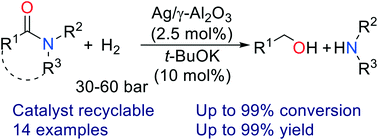
We report the heterogeneously catalyzed hydrogenation of amides to form alcohols and amines. This C–N bond cleavage reaction is catalyzed by silver on γ-alumina, proceeds smoothly in high yields and 100% selectivity, and is effective for both benzamides and aliphatic amides. This is in contrast to amide hydrogenation catalyzed by heterogeneous catalysts, which generally proceeds via C–O bond cleavage. Recycling experiments show that the catalyst Ag/γ-Al2O3 can be easily recovered and regenerated without decrease in catalytic activity.


 Please wait while we load your content...
Please wait while we load your content...