Influence of the nature of the support on the catalytic performance of CALB: experimental and theoretical evidence
Abstract
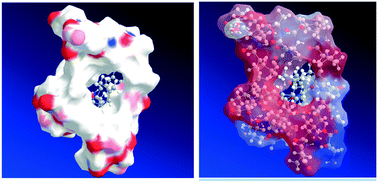
The immobilization of the lipase B of Candida antarctica (CALB) was carried out on various hydrophilic and hydrophobic supports through adsorption and covalent bond interactions. The catalytic performance of the biocatalysts was evaluated in the enantioselective esterification of R/S-ibuprofen with ethanol without co-solvents added. The present investigation demonstrates that the biocatalysts based on CALB immobilized by adsorption on hydrophobic supports (acrylic resin, polymethylmethacrylate, polystyrene, epoxy resin and polypropylene) exhibit higher conversion of ibuprofen and enantiomeric excess towards the S-enantiomer than CALB immobilized on hydrophilic materials. Additionally, the enzyme loading is a key factor influencing the conversion of the substrate regardless of the nature of the support material. A high enzyme loading is detrimental to the specific activity of the biocatalysts. Experimental and molecular modeling investigations provided evidence for the influence of water activity in the stereospecificity of the lipase.


 Please wait while we load your content...
Please wait while we load your content...