Visible light-driven selective hydrogenation of unsaturated aromatics in an aqueous solution by direct photocatalysis of Au nanoparticles†
Abstract
Selective hydrogenation of various chemical bonds, such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, C
C, C![[triple bond, length as m-dash]](https://www.rsc.org/images/entities/char_e002.gif) C, C
C, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, N
O, N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and C
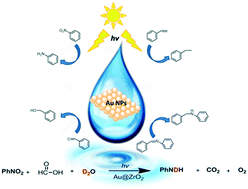
O, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, is efficiently driven by visible light over a supported gold nanoparticle (AuNP) photocatalyst under mild reaction conditions. The reaction system exhibits high substituent tolerance and tunable selectivity by light wavelength. Density functional theory (DFT) calculations demonstrated a strong chemisorption between the reactant molecule and metal resulting in hybridized orbitals. It is proposed that direct photoexcitation between hybridized orbitals is the main driving force of the hydrogenation reaction. The hydrogenation pathway is investigated by the isotope tracking technique. We revealed the cooperation of water and formic acid (FA) as a hydrogen source and the hydrogenation route through Au–H species on the AuNP surface.
N, is efficiently driven by visible light over a supported gold nanoparticle (AuNP) photocatalyst under mild reaction conditions. The reaction system exhibits high substituent tolerance and tunable selectivity by light wavelength. Density functional theory (DFT) calculations demonstrated a strong chemisorption between the reactant molecule and metal resulting in hybridized orbitals. It is proposed that direct photoexcitation between hybridized orbitals is the main driving force of the hydrogenation reaction. The hydrogenation pathway is investigated by the isotope tracking technique. We revealed the cooperation of water and formic acid (FA) as a hydrogen source and the hydrogenation route through Au–H species on the AuNP surface.


 Please wait while we load your content...
Please wait while we load your content...