The cooperation effect in the Au–Pd/LDH for promoting photocatalytic selective oxidation of benzyl alcohol†
Abstract
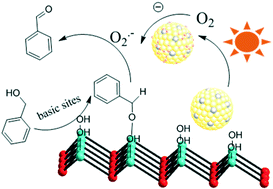
Materials with different Au–Pd ratios bimetallic alloy nanoparticles (NPs) supported on MgAl–LDH (Au–Pd/LDH) were prepared by an impregnation–reduction method and developed as photocatalysts for the selective oxidation of benzyl alcohol to benzaldehyde under visible light irradiation. A typical sample (Au9–Pd1/LDH) showed efficient photocatalytic activity with 91.1% conversion and 99% selectivity. The conversion was greatly increased from 3.6% for Au/LDH and 1.5% for Pd/LDH, respectively. Moreover, it was four times higher than that for Au9–Pd1/TiO2. The ESR spectrum demonstrated that the main reactive species were superoxide radicals (O2˙−) generated by the transfer of hot electrons from the Au–Pd alloy NPs to the surface adsorbed molecular oxygen. The formation of hot electrons was attributed to localized surface plasmon resonance (LSPR) of the alloy NPs. Based on XPS analysis, a synergistic electron effect in the Au–Pd alloy was observed due to the transfer of electrons from Pd to Au atoms. This effect enhances the adsorption ability of Pd atoms to oxygen molecules and facilitates the formation of O2˙−. The results from CO2-TPD and in situ FT-IR analyses indicated that benzyl alcohol could be efficiently activated at the surface base sites on the LDH via surface coordination to form a metal-alkoxide intermediate. Here, the intermediate could be more easily oxidized by O2˙− to form benzaldehyde. Finally, a possible mechanism based on the cooperation effect between the alloy NPs and the surface basic sites on the LDH was proposed to illustrate the photocatalytic process.


 Please wait while we load your content...
Please wait while we load your content...