Structural and chemical analysis of second-row impurities in liquid lead–bismuth eutectic by first-principles molecular dynamics†
Abstract
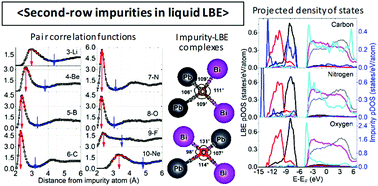
The structural and chemical states of the second-row impurities in liquid lead–bismuth eutectic (LBE) are studied by first-principles molecular dynamics. First, several structural quantities such as the number of first-neighboring atoms and the LBE–impurity–LBE characteristic angle are obtained to determine the impurity–LBE local structure. Next, the impurity charge states and the electronic density of states are analyzed to reveal the chemical states of the impurities in the liquid LBE. It is observed that for the majority of impurities the 2p–6p interaction specifies the local structure around the impurity as well as the chemical state of the impurity. The anisotropy in the 2p–6p covalent interaction causes the tetrahedron-wise structures for B, C, N, and O, and the ionic interaction is relatively strong for Li and F. The change in the interaction scheme over the second-row impurities can be explained from the downward shift of the 2s and 2p orbital energy levels as the atomic number increases. Further, some impurities indicate interaction preferences for either Pb or Bi. These findings can assist the understanding and prediction of the behavior of impurity atoms in liquid LBE.


 Please wait while we load your content...
Please wait while we load your content...