Femtosecond infrared spectroscopy reveals the primary events of the ferrioxalate actinometer†
Abstract
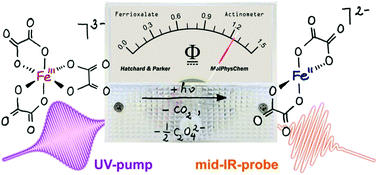
Chemical actinometry is an indispensable analytical tool in preparative photochemistry that allows for a precise measurement of radiant fluxes inside photoreactors. An actinometer thus enables an absolute determination of the quantum yield of a photochemical reaction of interest. The “gold standard” of chemical actinometry in liquid systems is the Hatchard–Parker actinometer, i.e. an aqueous solution of potassium trisoxalatoferrate(III), which is based on the light-induced net transformation of ferric into ferrous oxalate complexes. Although the absolute photochemical quantum yield for this fundamental standard system has been accurately known for many years, the underlying molecular-level mechanisms and time scales associated with a photoreduction of the ferrioxalate actinometer remained so far largely obscured. Here, we use femtosecond mid-infrared spectroscopy combined with ultrafast laser photolysis to obtain unique structural-dynamical information associated with the primary light-triggered processes thereby finally providing the missing quantitative molecular-level foundations that ultimately justify a utilization of aqueous ferrioxalate as a true photochemical standard. Following photon absorption by the octahedral parent complex, an ultrafast decarboxylation occurs within 500 fs, which generates a penta-coordinated ferrous dioxalate that carries a bent carbon dioxide radical anion ligand in an “end-on” O-coordinated fashion. This unique intermediate structurally isomerizes on a tens of picoseconds time scale and subsequently loses a CO2˙−-ligand to form a square-planar bisoxalatoferrate(II) on a hundreds of picoseconds time scale.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...