Atomically-defined model catalysts in ultrahigh vacuum and in liquid electrolytes: particle size-dependent CO adsorption on Pt nanoparticles on ordered Co3O4(111) films†
Abstract
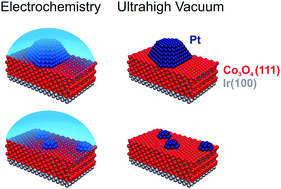
We have studied particle size effects on atomically-defined model catalysts both in ultrahigh vacuum (UHV) and under electrochemical (EC) conditions in liquid electrolytes. The model catalysts were prepared in UHV by physical vapour deposition (PVD) of Pt onto an ordered Co3O4(111) film on Ir(100), yielding nanoparticles (NPs) with an average size from 10 to 500 atoms per particle (0.8 to 3 nm). The model systems were characterized in UHV using surface science methods including scanning tunnelling microscopy (STM), before transferring them out of the UHV and into the electrolyte without contact to ambient conditions. By X-ray photoelectron spectroscopy (XPS) we show that the model surfaces are stable in the EC environment under the applied conditions (0.1 to 1 M phosphate buffer, pH 10, 0.33 to 1.03 VRHE). As a reference, we study Pt(111) under identical conditions. In UHV, we also investigated the adsorption of CO using infrared reflection absorption spectroscopy (IRRAS). Under EC conditions, we performed equivalent experiments using EC infrared reflection absorption spectroscopy (EC-IRRAS) in combination with cyclic voltammetry (CV). Characteristic differences were observed between the IR spectra under EC conditions and in UHV. Besides the red-shift induced by the interfacial electric field (Stark effect), the EC IR bands of CO on Pt(111) show a larger width (by a factor of 2) as a result of local variations in the CO environment and coupling to the electrolyte. The CO IR bands of the Pt NPs are even broader (by a factor of 5), which is attributed to local variations of the interfacial electric field at the NP surface. Further pronounced differences are observed between the spectra taken in UHV and in the electrolyte regarding the site occupation and its dependence on particle size. In UHV, adsorption at on-top sites is preferred on Pt(111) at low coverage and similar adsorption ratios of on-top and bridge-bonded CO are formed at saturation coverage. In sharp contrast, on-top adsorption of CO on Pt(111) is partially suppressed under EC conditions. This effect is attributed to the competitive adsorption of anions from the electrolyte and leads to a clear preference for bridge sites at higher potentials (>0.5 VRHE). For the Pt NPs, the situation is different and an increasing fraction of on-top CO is observed with decreasing particle size, both under EC conditions and in UHV. For the smallest particles (10–20 atoms) we do not detect any bridge-bonded CO. This change in site preference as a function of particle size is attributed to stronger on-top adsorption on low-coordinated Pt atoms of small Pt NPs. The effect leads to a clear preference for on-top adsorption in the electrolyte even at low CO coverage and over the full potential range studied.


 Please wait while we load your content...
Please wait while we load your content...