Water-mediated network in the resistance mechanism of fosfomycin†
Abstract
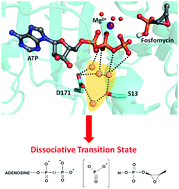
Fosfomycin Resistance Kinase A (FomA) catalyzes the phosphorylation of fosfomycin, which is an effective antibiotic for treating urinary tract infections. Understanding the chemical reaction mechanism is essential for developing strategies to counter the resistance of fosfomycin in clinical settings. Here the catalytic mechanism of FomA was investigated using molecular dynamic simulations in conjunction with quantum mechanics/molecular mechanics calculations (B97d/AMBER99). Our QM/MM study disclosed that the phosphorylation reaction catalyzed by FomA follows a dissociative mechanism, in contrast to the previously proposed associative mechanism. In addition, we found that His58, a characteristic residue in the AAK family, plays a key role in positioning the phosphate group of fosfomycin in the transition state. Molecular dynamic simulations revealed the important roles of Lys9 and Lys18 in arranging the nucleotide for phosphate transfer. Furthermore, we identified a four-membered water network mediated by Asp171 and Ser13 that is critical in ordering ATP for phosphate transfer. The active structure and reaction mechanism of FomA will provide valuable insights for developing new strategies to tackle the resistance to Fosfomycin-based antibiotic therapies.


 Please wait while we load your content...
Please wait while we load your content...