Flue gas adsorption on periodic mesoporous phenylene-silica: a DFT approach†
Abstract
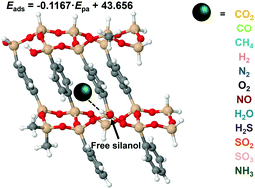
Periodic mesoporous organosilicas (PMOs) were suggested as potential adsorbents for CO2/CH4 separation because of their large affinities towards CO2 and low interaction with CH4. Herewith, we present a comprehensive computational study on the binding properties of flue gas species with the pore walls of periodic mesoporous phenylene-silica (Ph-PMO) for understanding the possible impact of other gaseous species in the CO2/CH4 separation. The calculations considered three exchange–correlation functionals (PBE, PBE-D2 and M06-2X) based on the density functional theory and the walls of the periodic mesoporous phenylene-silica were modelled within the cluster model approach. The components of the flue gas considered were the diatomic CO, H2, N2, O2 and NO molecules, the triatomic CO2, H2O, H2S and SO2 species, the tetratomic SO3 and NH3 gases and the pentatomic CH4 molecule. The calculated data demonstrate that the presence of H2O, SO2, NH3, H2S and SO3 is a significant threat to CO2 capture by Ph-PMO and suggest that the Ph-PMO material would present high selectivity for CO2 over CH4, CO, H2 or N2 adsorption. The adsorption behaviour of flue gas components in Ph-PMO can be directly related to the experimental proton affinities, basicities or even the polarizabilities of the gaseous molecules.


 Please wait while we load your content...
Please wait while we load your content...