Mechanism of enhanced triplet decay of thionucleobase by glycosylation and rate-modulating strategies†
Abstract
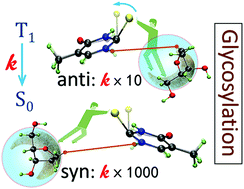
The decay of the triplet state of photosensitizers is essential to their performance in singlet-oxygen generation. Experiments have shown that in thionucleosides, this decay is enhanced compared to that in the corresponding thionucleobases. In this work, we applied quantum-chemical methods and chemical-kinetic modeling to investigate the effects of the sugar substituent on the triplet decay of thionucleosides. The computed rates for the energetically favored conformers of thiothymidine, thiouridine, and thioguanosine (and the respective thionucleobases) show a remarkable quantitative agreement with the experimental results. We additionally show that the triplet decay enhancement is caused by the repulsion interaction between the sugar group and the sulfur atom, which reduces the activation energy for intersystem crossing by destabilizing the T1 minimum. In some instances, an intramolecular hydrogen bond stabilizes the energy of the T1/S0 crossing point, also reducing the activation energy. This molecular understanding of the mechanism of enhanced triplet decay provides a guideline to control the triplet decay rate, which was tested in new thiothymidine derivatives.
- This article is part of the themed collection: 2018 PCCP HOT Articles


 Please wait while we load your content...
Please wait while we load your content...